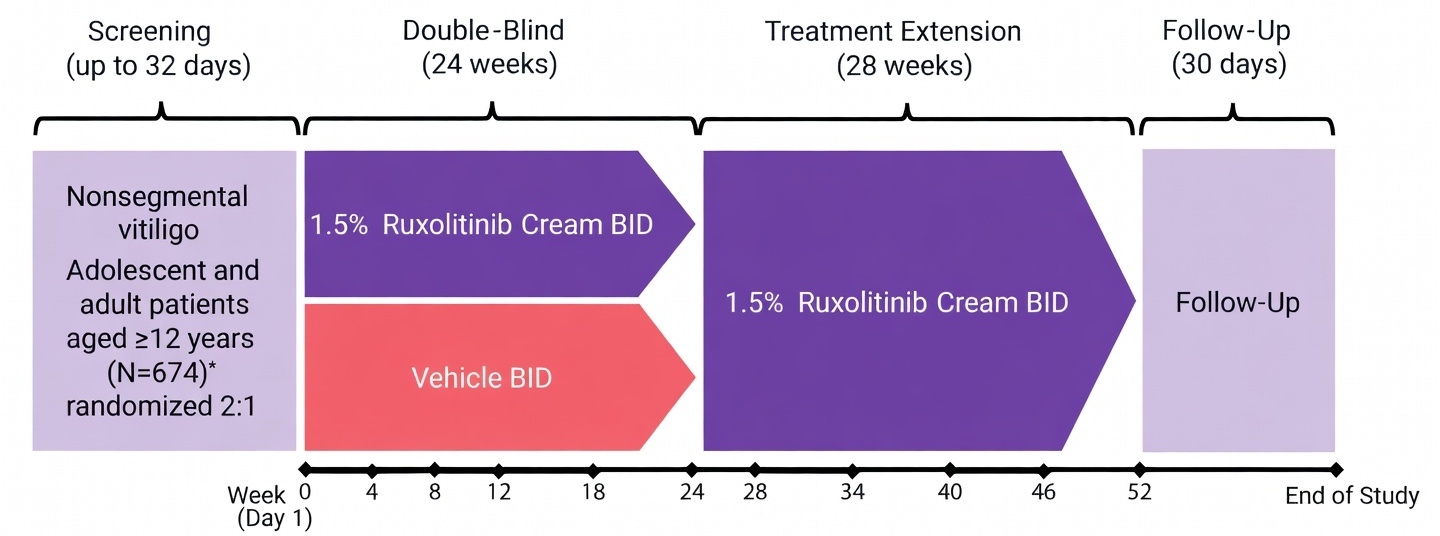
Lumirix® Efficacy: Phase 3 Clinical Evidence
This image is for illustrative purposes only.

This image is for illustrative purposes only.
*1 randomized patient who did not apply ≥1dose of Ruxolitinib cream was excluded from safety analyses.
13 patients from 1 study site were excluded from efficacy analyses for compliance issues.
% of patients achieving an F-VASI75 response at week 24.²
% of patients achieving F-VASI50 and F-VASI90 responses.
% of patients achieving a T-VASI50 response.
% of patients achieving a VNS response of 4 (‘a lot less noticeable’) or 5 (‘no longer noticeable’).
% change from baseline in affected F-BSA.
Safety and tolerability.
% change from baseline in F-VASI, T-VASI, affected F-BSA and T-BSA during the treatment period.
% of patients having F-VASI improvements or T-VASI improvements.
The face includes the area on the forehead to the original hairline, on the cheek to the jawline vertically and laterally from the corner of the mouth to the tragus. It includes the surface area of the nose but not that of the lips, scalp, eyelids, ears, or neck.² VNS was assessed for facial lesions only.²
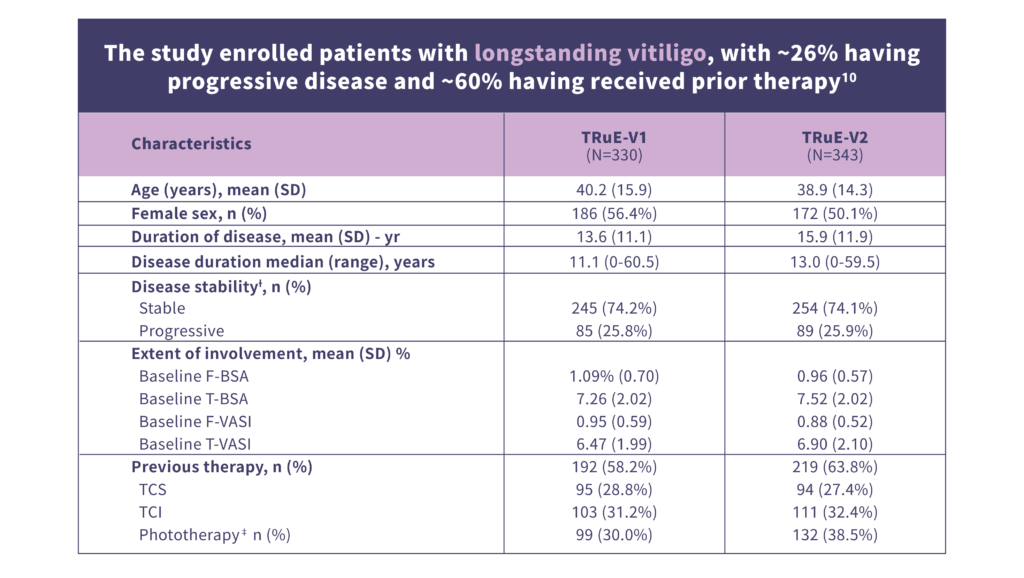
ƗDetermination of disease stability was based on the investigator’s judgment.
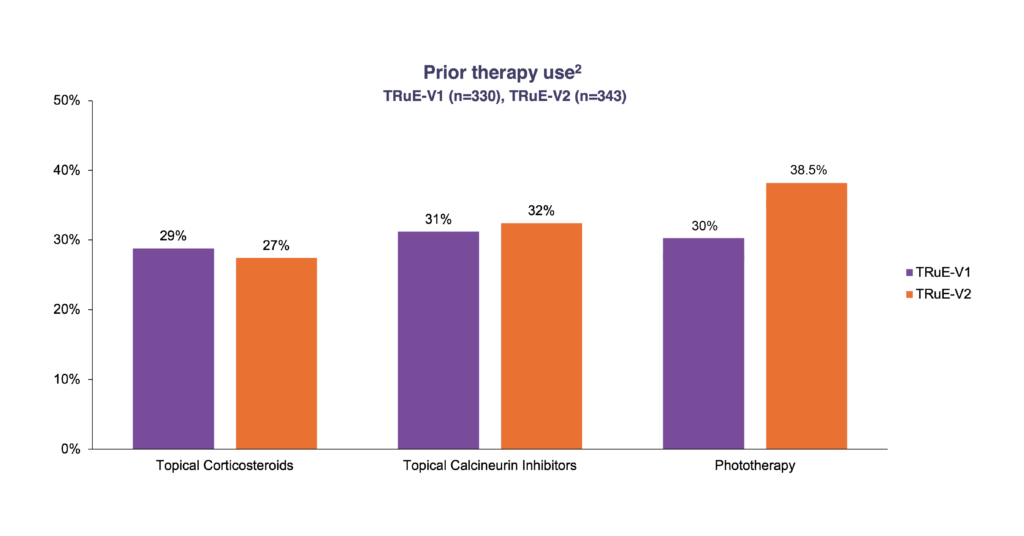
ǂ Patients could have used multiple previous lines of therapy.
Efficacy of Ruxolitinib cream in Achieving the Primary Endpoint of F‑VASI75 Response.
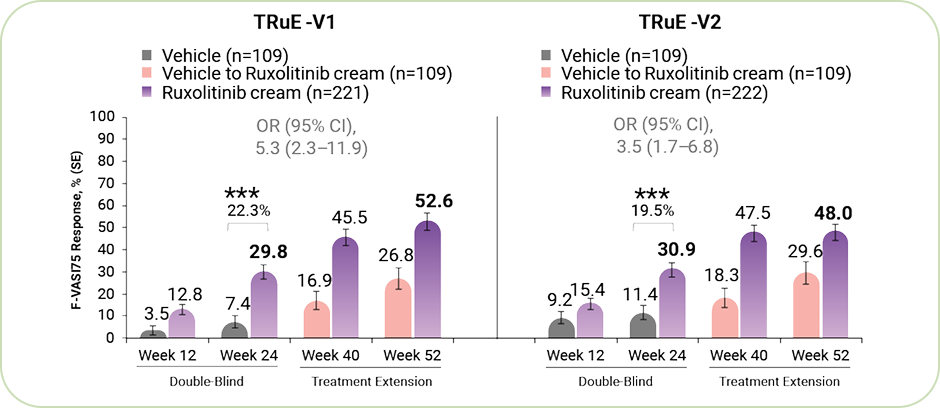
*** P<0.001 for response rate difference for ruxolitinib cream vs vehicle.
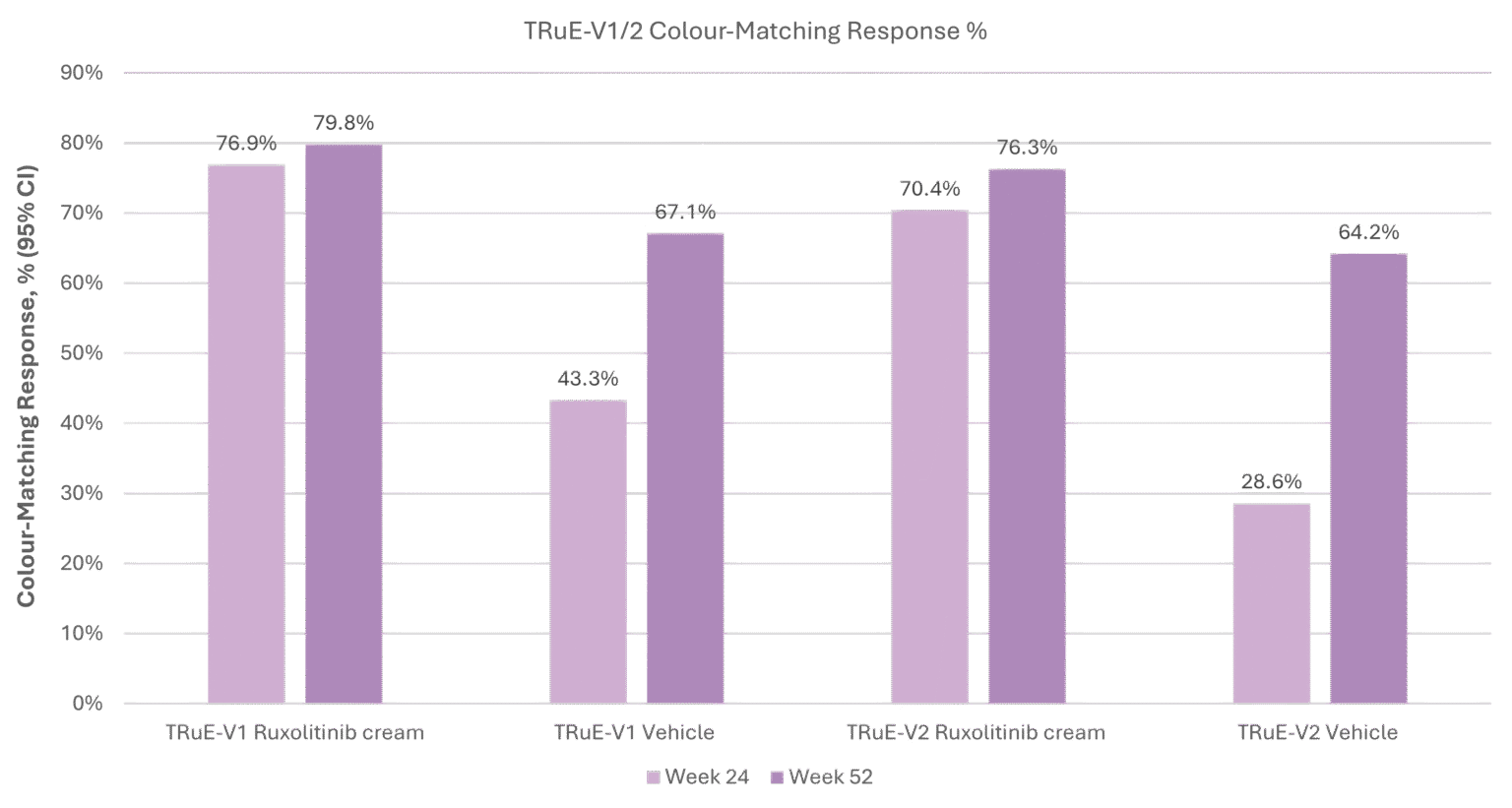
§Color-matching response was defined as achieving a rating of good, very good, or excellent.
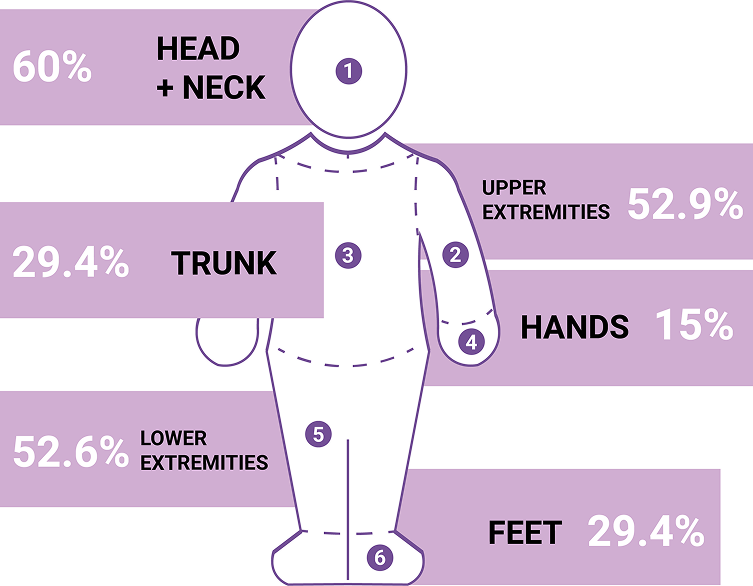
Adapted from ref. 11
Long‑term efficacy of ruxolitinib cream
Durability of repigmentation
Relapse rates after stopping treatment
Safety over long‑term continuous use
Continued long‑term responses in patients with limited or no early repigmentation at Week 24
At Week 52 of the parent studies, patients were assigned to one of two cohorts based on their facial response:
These were the high responders who achieved ≥90% facial repigmentation.
This group allowed researchers to evaluate whether repigmentation can be maintained without continuous therapy.
Primary end point : Time to relapse (defined as a <F-VASI75 response) after treatment discontinuation among patients randomised to vehicle.
Secondary end point : Time to maintain a ≥F-VASI90 response after treatment discontinuation.
These were patients who still had room for improvement.
They continued open‑label ruxolitinib cream to measure additional gains in repigmentation over time.
End point : Shifts and maintenance of facial and total body repigmentation
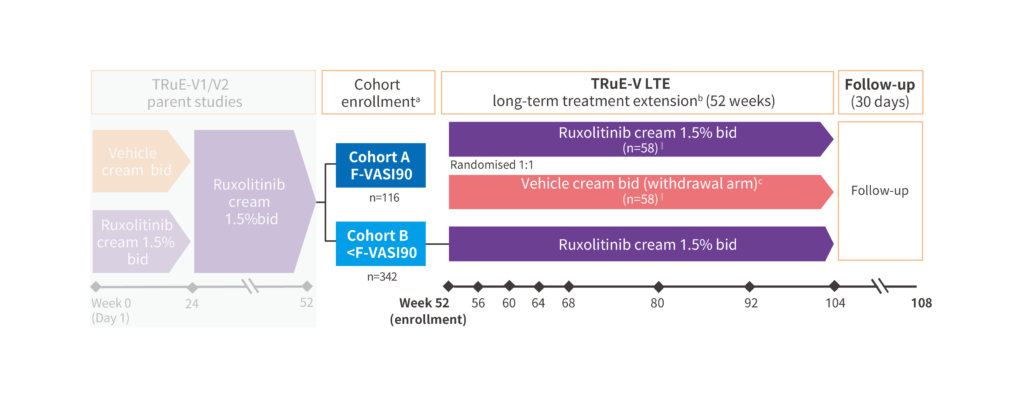
a Cohort enrollment was based on clinical response at week 52 of parent studies.
b Concomitant vitiligo treatments, such as phototherapy, were not permitted during the TRuE-V LTE study.
cPatients randomised to vehicle who relapsed (<F-VASI75) could apply Ruxolitinib cream 1.5% BID rescue treatment for the remainder of the LTE period.
‖ Two patients (n=1 from each arm) were excluded from the analysis due to protocol nonadherence at the site. Three patients were incorrectly assigned to Cohort A (treatment-continuation arm, n=2; treatment-withdrawal arm, n=1) and included in the analyses.
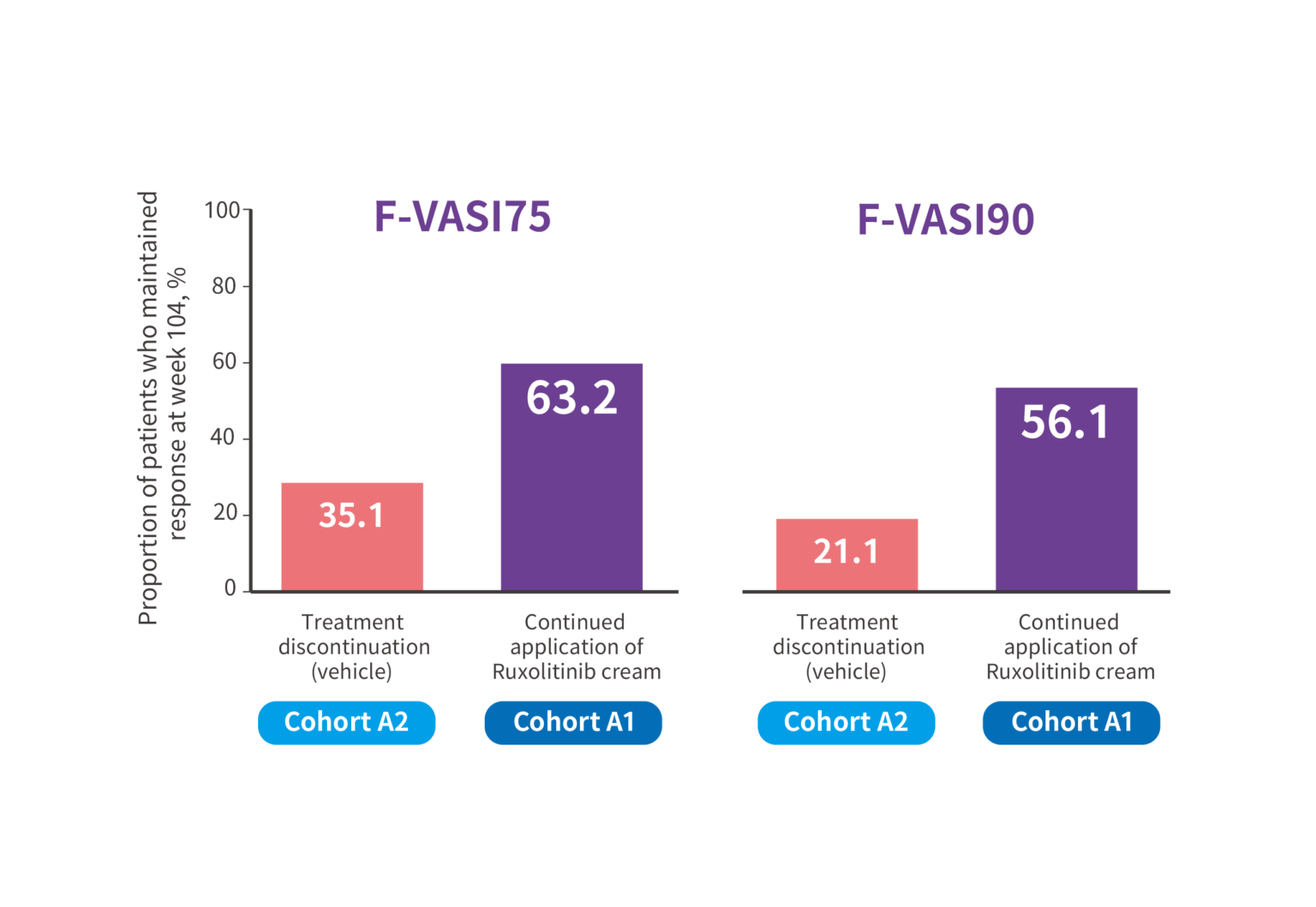
Continued application of ruxolitinib cream; n=57; treatment discontinuation (vehicle); n=57
At week 104, over 63% of patients who continued application of Ruxolitinib cream maintained F-VASI75, and over 56% maintained
F-VASI90 respectively.
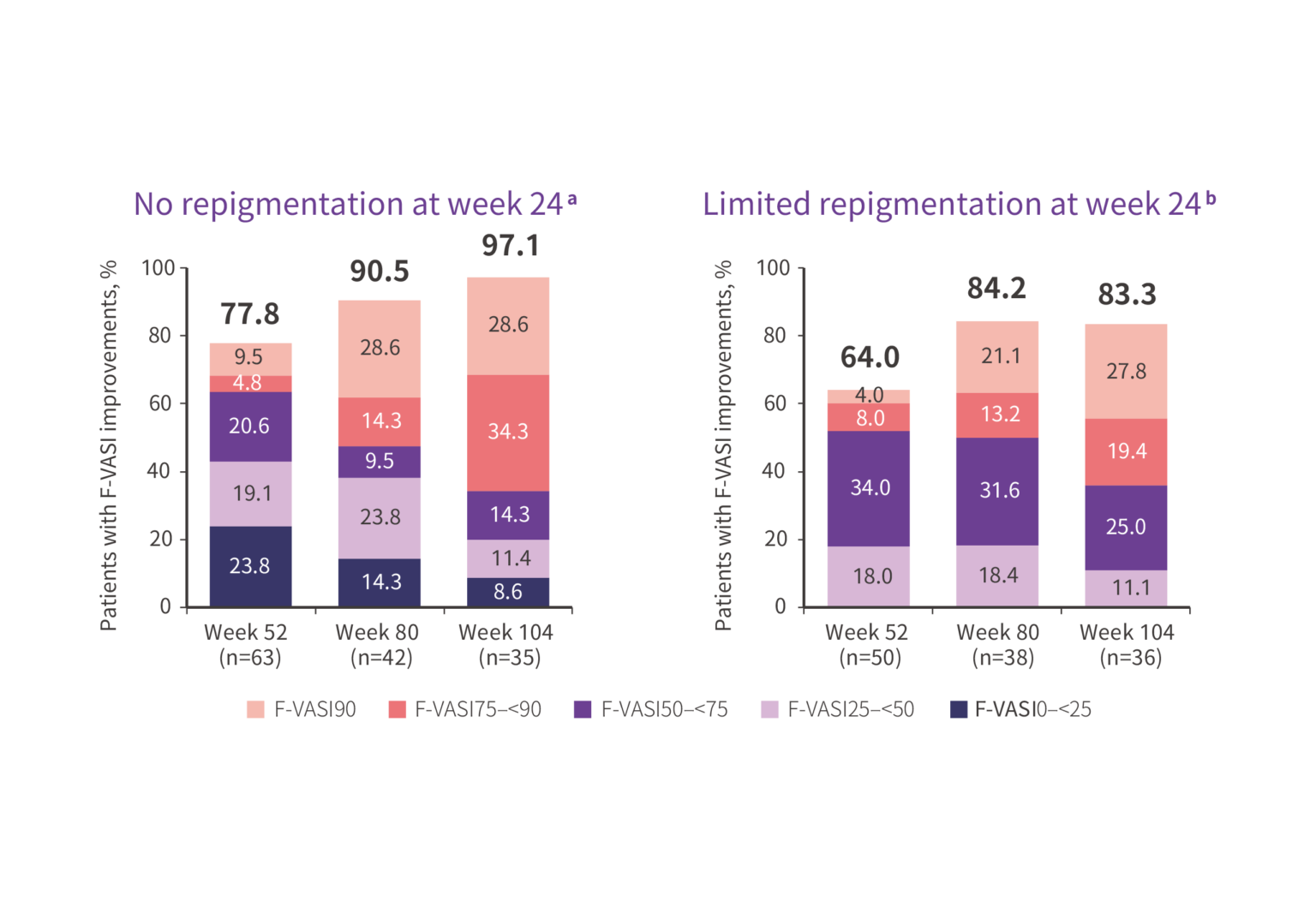
Data at weeks 80 and 104 are from cohort B.
a Patients with worsening or no improvement in F-VASI (ie, ≤0%) at week 24 and nonmissing F-VASI values at weeks 52, 80, or 104.
b Patients with >0%–<25% improvement in F-VASI at week 24 and nonmissing F-VASI values at weeks 52, 80, or 104.
At week 104, more than 8 out of 10 participants from Cohort B gained
F-VASI improvements.
F-BSA: facial body surface area; NB-UVB: narrow-band ultraviolet-B; PUVA: psoralen ultraviolet-A; T-BSA: total body surface area.
Are you a healthcare professional practising in Hong Kong and Macau?