Look into the Safety Data
This image is for illustrative purposes only.

This image is for illustrative purposes only.
The safety profile of Lumirix® is established in Phase 3 studies with up to 104 weeks of exposure.10,16,17
At 52 weeks, Lumirix® was generally well-tolerated. The majority of application‑site reactions were mild and
transient. Serious treatment‑related adverse events were infrequent.
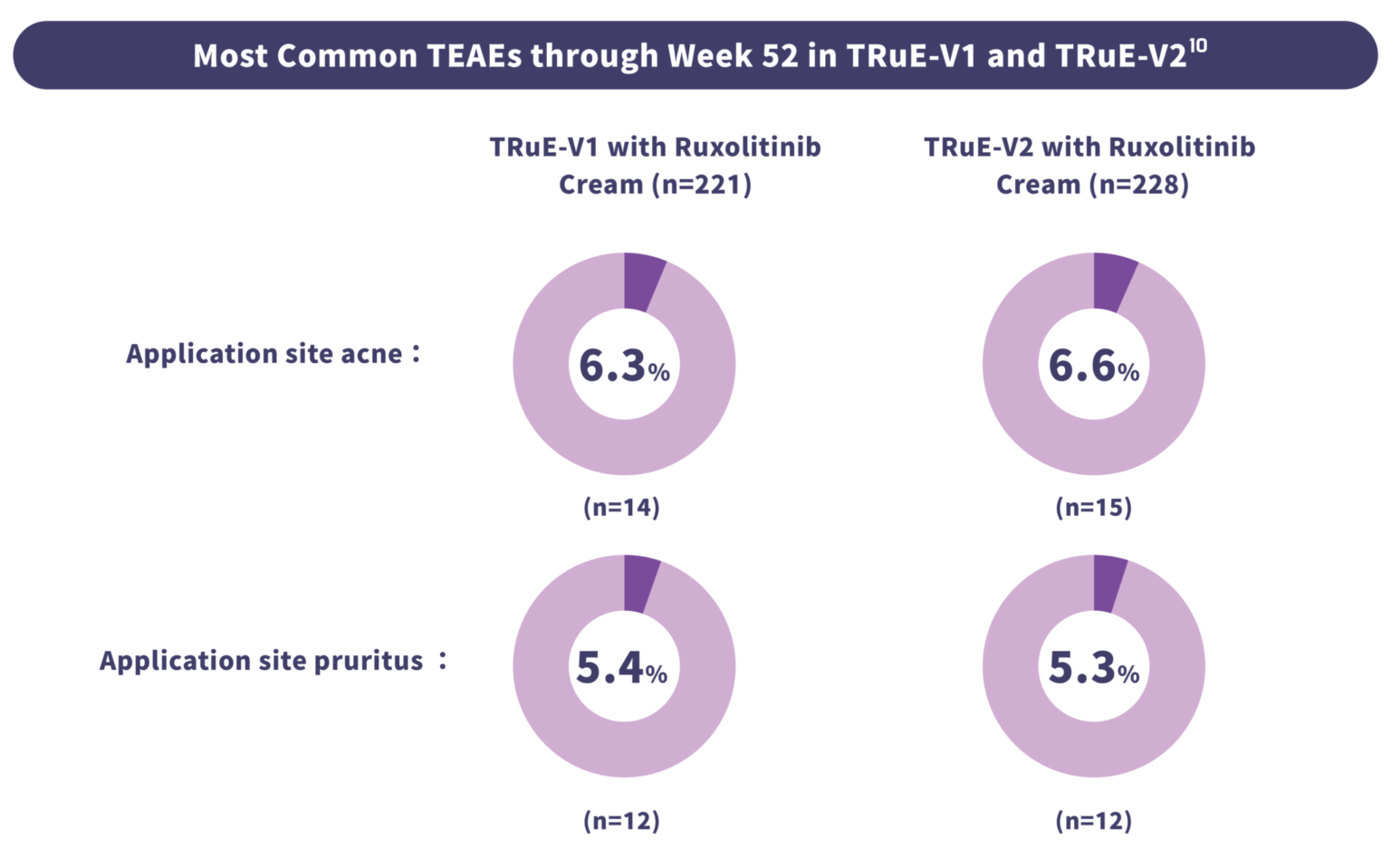
No clinically significant application site reactions or serious treatment-related AEs.2

*Occurring in ≥4% of patients in any treatment group. †No serious TEAEs were considered rested to trestment.
Ruxolitinib cream 104-week safety profile was consistent with TRuE-V1/V2 (52 weeks) and no new safety
signals were reported.16,17
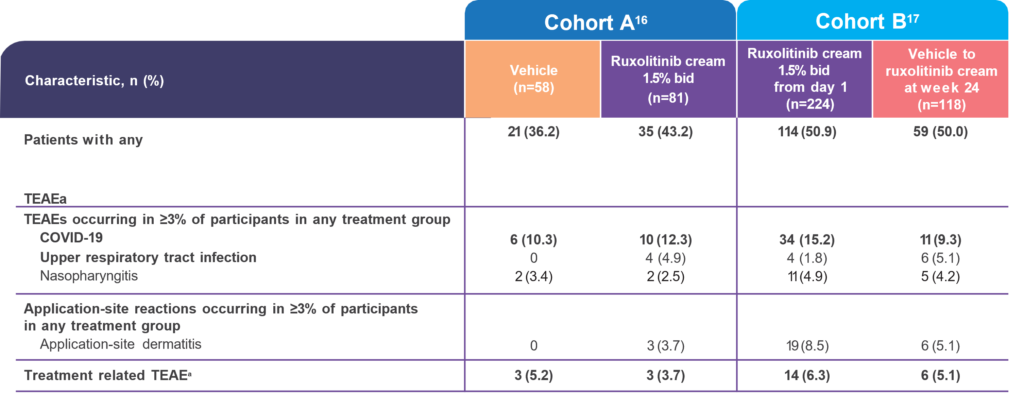
Ruxolitinib cream from day 1 and vehicle to ruxolitinib cream at week 24 indicate the treatment arms of the TRuE-V LTE study.
a Treatment-related TEAEs in patients applying ruxolitinib cream 1.5% bid were application-site dermatitis (n=1), application-site rash (n=1), and hyperlipidemia (n=1) in cohort A and application-site pruritus (n=4; 1.2%) in cohort B.2
Are you a healthcare professional practising in Hong Kong and Macau?